rms Co. Scales Medical Device Production With New 3D Printer
A large US-based medical device manufacturer adds DMP Flex 350 Dual to its fleet of more than 30 3D Systems printers.

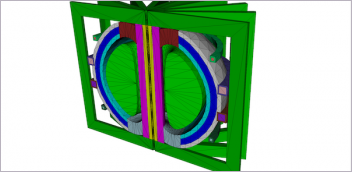
3D Systems has helped rms expand its portfolio of direct metal printers from one machine to more than 30, enabling scaled production. Image courtesy of 3D Systems.
Latest News
January 19, 2023
3D Systems reports that rms Co., a large contract manufacturer of tight-tolerance medical devices in the world, has added the DMP Flex 350 Dual to its production workflow. rms has more than 30 3D Systems’ DMP 3D printers in its additive manufacturing facility, enabling the company to provide a complete Powder to Package capability to medical device OEMs, according to 3D Systems.
Powder to Package encompasses the entire manufacturing and post-processing workflow required to go from a digital file to a sterilized and packaged final product ready for the surgical suite. With the addition of the DMP Flex 350 Dual, rms believes they will be able to expand the types of implants they produce and accelerate throughput.
In 4 years, 3D Systems has helped rms expand its portfolio of direct metal printers from one machine to more than 30, enabling scaled production. As a result, rms has been able to cement its ability to additively manufacture titanium implants. By adding the DMP Flex 350 Dual, rms will be able to increase throughput for smaller implants including those intended for spinal applications. They believe the same productivity increases will also open opportunities in the production of large joint and extremity implants while allowing flexibility for existing projects.
“The increase in efficiency we get from the DMP Flex 350 Dual system creates new opportunities for larger and taller parts which were economically challenging with a single laser system,” says Ryan Kircher, senior additive manufacturing engineer, rms Company. “Another key reason rms Company decided to invest in a DMP Flex 350 Dual is that it is engineered in a way that allows us to produce the high precision, high-quality products we expect from our current installation of DMP systems without changing the processing parameters we validated when we initially developed our AM capabilities.”
“Our Powder to Package solution provides a complete end-to-end value proposition that allows us to control the entire manufacturing and packaging process flow,” says Troy Olson, director of operations—additive manufacturing division, rms Company. “A key element of our process flow is the reliability and performance we get from our stable of 30-plus 3D Systems’ DMP ProX 320 and DMP Flex 350 machines. During our validation process, the DMP Flex 350 Dual showed no measurable differences in mechanical properties or dimensional accuracy. As we continue to scale our additive manufacturing business, the 3D Systems DMP Flex 350 Dual will be at the forefront of our growth strategy.”
“Our Application Innovation Group worked alongside the rms team to develop, characterize and validate the DMP Flex 350 Dual as a seamless solution within their production environment,” sysd Jeph Ruppert, director, application innovation group, 3D Systems. “Our collective application and technology expertise combined with the unparalleled capabilities of the DMP Flex 350 Dual is helping drive innovation for rms.”
“Our collaboration with rms has spanned several years, and during that time rms has grown at a rate and scale that is truly impressive,” says Menno Ellis, executive vice president, healthcare solutions, 3D Systems. “They have not only delivered excellent solutions to the medical device community but have also grown their in-house additive knowledge, helping guide the industry to a more mature and robust position.”
Sources: Press materials received from the company and additional information gleaned from the company’s website.
More 3D Systems Coverage
Subscribe to our FREE magazine, FREE email newsletters or both!
Latest News
About the Author
DE’s editors contribute news and new product announcements to Digital Engineering.
Press releases may be sent to them via DE-Editors@digitaleng.news.